1
Introduction
Surface modifications that introduce fluorinated compounds (–CF₂– and –CF₃ groups) with low surface energy are conventionally used enhance the performance of water-repellent and antismudge coatings. A representative class of such chemicals is per- and polyfluoroalkyl substances (PFAS), which have been used in applications such as cookware and antismudge coatings for displays.
However, in recent years, PFAS have become strongly recognized internationally as “forever chemicals” that are difficult to degrade and remain in the environment over a long term. This raises concerns regarding their toxicity and environmental burden. However, if the use of PFAS is banned, water repellency, antismudge, and oil repellency of products will be lost, and their lifespan will be shortened. Simultaneously, alternative PFAS technologies face challenges in terms of achieving high performance, durability, and cost; hence, more practical alternatives are needed.
As regulations and phased bans progress across various countries, including European countries, the United States, and Japan, the establishment of PFAS-independent water-repellent and antismudge technologies has become urgent. To date, two alternative approaches have been explored: (1) the formation of chemically water-repellent films using fluorine-free low-surface-energy materials1) and (2) structural water repellency, which reduces the liquid–solid contact area through micro- or nanostructures on the surface2).
Considering that the water-repellent and antismudge performances vary based on the application, substrate, and required durability, designing and optimizing the microstructures and coating materials in an integrated manner is necessary. However, most studies have focused on either microstructure design via microfabrication or chemical surface modification alone, and data enabling the integrated optimization of both is insufficient.
Therefore, in this study, a practical PFAS-free water-repellent technology was established. Specifically, we adopted structural water repellency as a basic policy without excessive reliance on chemical surface modification by combining silicone-based and alkyl-based chemical surface modification to establish.
2
Experimental section
A key challenge in designing PFAS-free water-repellent systems is the difficulty of achieving water repellency levels similar to those shown by conventional fluorinated water repellents by relying solely on chemically low surface energy. Therefore, in this study, we focused on the lotus effect (stabilization of the Cassie–Baxter state) that originates from fine surface textures and developed a method to express water repellency comparable with that of fluorinated materials without using PFAS.
2-1. Water-Repellent Material Surface Energy Measurements
Wettability is an important indicator of water-repellency. Wettability indicates how easily a liquid spreads on a solid surface and is governed by the interaction between the surface free energy of the solid and the surface tension of the liquid. When a liquid droplet is placed on a solid surface, three interfaces coexist simultaneously: solid–gas (sv), solid–liquid (sl), and liquid–gas (lv). The droplet shape (contact angle θ) is determined such that the interfacial free energies are minimized and is described by Young’s equation as follows:
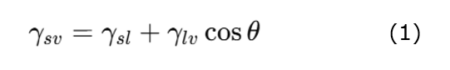
Furthermore, according to the Young–Dupré equation, the work of adhesion (Wsl) is expressed as follows:
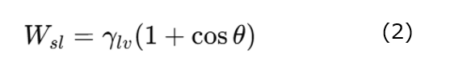
Thus, the contact-angle θ reflects the energy state of the solid surface.
In this experiment, we decomposed the surface free energy into components (dispersive, dipolar, and hydrogen-bonded components). We used Teflon (PTFE) as a representative fluorinated material and silicone- and hydrocarbon-based materials as non-fluorinated materials. The contact angles were measured using three solvents (n-hexadecane, diiodomethane, and water), and the surface free energies were calculated by applying the extended Fowkes theory. The measured contact angles and calculated surface free energies are listed in Table 1.
Table 1 Components of Surface Free Energy for Various Materials
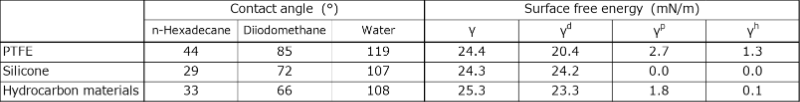
Although the surface free energies of fluorinated and non-fluorinated materials were comparable, PTFE exhibited superior contact angles for each solvent. This suggests that the contact angle was strongly influenced by the dispersive component. Given that the dispersive component is determined by the chemical structure, achieving a surface free energy equivalent to that of fluorinated materials using nonfluorinated materials was limited.
Accordingly, we shifted our approach to improving water repellency through structural design. To examine combinations of surface textures and material properties, we focused on lotus leaves, which exhibit high superhydrophobicity in nature. Lotus leaves have micrometer-scale protrusions of the order of several tens of micrometers. These protrusions are covered by a wax layer, and this structure forms an air layer that confers superhydrophobicity. In this study, we mimicked the lotus-leaf structure by forming textures ranging from submicron to several tens of micrometers and investigated the conditions for achieving water and oil repellency by combining these textures with non-fluorinated materials.
2-2. Fabrication of Surface Microstructures
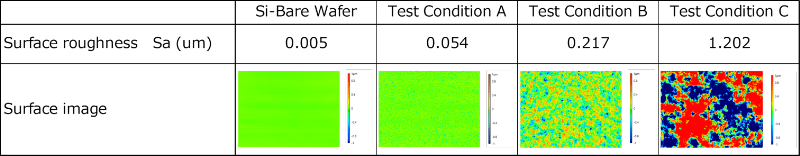
We employed blasting as a fabrication method to realize the targeted surface textures. Briefly, a silicon wafer (t = 0.5 mm) was used as the substrate. Blasting was performed using a pressure-feed blasting apparatus, and alumina (#320 to #2000) was used as the blasting medium. Samples with different surface roughness values were prepared by adjusting the blasting conditions. The measured surface roughness values of the fabricated samples are listed in Table 2. The surface roughness was measured using a laser microscope.
Table 2 Surface Roughness and Topographical Features
3
Results and discussion
To verify whether the intended effect was achieved, we coated the textured structures with a non-fluorinated material (silicone) and evaluated their water and oil repellency. To clarify the relationship between the surface roughness and wettability, we measured the contact angles of water and oil (oleic acid) on samples whose roughness was controlled stepwise by blasting. Figure 1 shows the relationship between the surface roughness and contact angle.
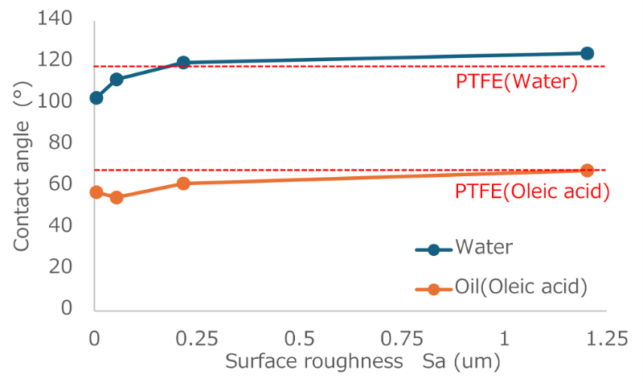
Fig. 1 Effect of surface roughness on contact angle after coating with silicone materials.
The water contact angle tended to increase with increasing Sa. Specifically, although the contact angle on the untreated flat surface (Sa = 0.005 µm) was 102.9°, it reached 124.4° on the textured surface with Sa = 1.20 µm. This is because the textured surface reduces the solid–liquid contact area and produces a Cassie–Baxter state mediated by an air layer.
In contrast, the contact angle of oleic acid was generally lower than that of water, and the improvement with increasing Sa was limited. The contact angle on the flat surface was 57.3° and 67.8° on the textured surface with Sa = 1.20 µm. Hence, a contact angle comparable with that of the fluorinated material (PTFE) was achieved using a non-fluorinated material (silicone). However, these results also indicated a large difference between water and oleic acid in terms of contact angle improvement.
Based on the measured results, we calculated the apparent fractional contact area ratio between the solvent and the solid surface using the Cassie–Baxter theoretical equation. Figure 2 shows the calculated fractional contact areas for water and oil (oleic acid) as a function of surface roughness.
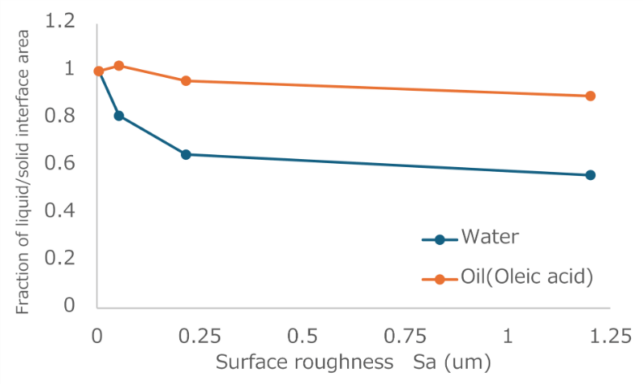
Fig. 2 Relationship between surface roughness and fractional surface area
The fractional contact area for water decreased as surface roughness increased, reaching a value as low as 0.56 on the textured surface with Sa = 1.20 µm. In contrast, for oleic acid, the fractional contact area remained as high as 0.90 even on the textured surface with Sa = 1.20 µm. This was attributed to oleic acid having a lower surface tension than that of water, rendering it easier for the liquid to penetrate and more difficult to maintain the Cassie state.
This study demonstrates that a combination of surface textures and a non-fluorinated material (silicone) can realize water and oil repellency comparable with that of fluorinated materials (PTFE). Simultaneously, the effect of texture was not sufficiently expressed for the oil (oleic acid). Therefore, adopting structures that inhibit droplet penetration is necessary to effectively express the structural effects against liquids, such as oils and organic solvents, with even lower surface free energies. Candidate structures include fractal and reentrant (inverse-taper) structures. Accordingly, in the future, we will establish the optimal structural design and further improve the liquid repellency.
4
Conclusion
In response to the strengthening of PFAS regulations, this study aimed to establish a water-repellent technology that did not rely on fluorinated materials, such as PFAS, by investigating combinations of microstructure design and fluorine-free low-surface-energy materials. By examining the microstructures and surface coating materials, we identified the structural conditions that stably form and maintain the Cassie–Baxter state, as well as a synergistic effect with surface-energy reduction induced by silicone materials. Consequently, we achieved a high level of water repellency comparable with that of the fluorinated material (PTFE) without using PFAS.
These results demonstrate the effectiveness of optimizing both “structure” and “chemistry,” which are two elements that have often been studied separately. Moreover, the study findings provide important insights toward simultaneously reducing environmental burden and achieving high functionality.
Furthermore, this technology offers high flexibility in material selection and applicable substrates and is expected to be deployed across a wide range of materials, including optical components, sensors, display covers, and medical devices. To develop a more practical surface technology in the future, we will amplify the microfabrication process for mass production, evaluate its durability, and enhance multifunctional integration with properties such as oil repellency and antismudge performance.



